The first and only TAVR system FDA-approved
for aortic regurgitation
Watch the full late-breaking
presentation from TCT
(n=180)
(n=180)
(n=141)
(n=700)
(n=700)
(n=360)
Primary 30-day safety endpoint† met
The Trilogy valve met a prespecified non-inferiority performance goal based on historical data (p < 0.0001).
| Variable | Pivotal (n=180) |
Pivotal + CAP (n=700) |
|---|---|---|
| All Cause Mortality | 2.2% | 1.6% |
| Stroke | 2.2% | 1.7% |
| Major Life-Threatening Bleeding | 4.4% | 2.7% |
| Acute Kidney Injury Stage 2 or 3 (7 Days) | 1.1% | 0.7% |
| Major Vascular Complication | 3.9% | 2.4% |
| Surgery, Intervention, or Major Vascular Complication Related to Device or Procedure | 2.8% | 3.0% |
| New Permanent Pacemaker Implantation※§ | 24.0% | 21.6% |
| Moderate or Severe Total Aortic Regurgitation¶ | 0.6% | 0.5% |
| Total | 26.7% | 27.0% |
Primary 1-year efficacy endpoint‡ met
Untreated severe, symptomatic aortic regurgitation is associated with high mortality. The Trilogy valve met a prespecified non-inferiority performance goal comparing treatment with Trilogy TAVR to conservative management (p < 0.0001).
(n=180)
(n=500)
Lower rates of new pacemaker observed toward the end of the trial
New pacemaker rates decreased with changes to procedural technique, sizing, and management of periprocedural conduction abnormalities.

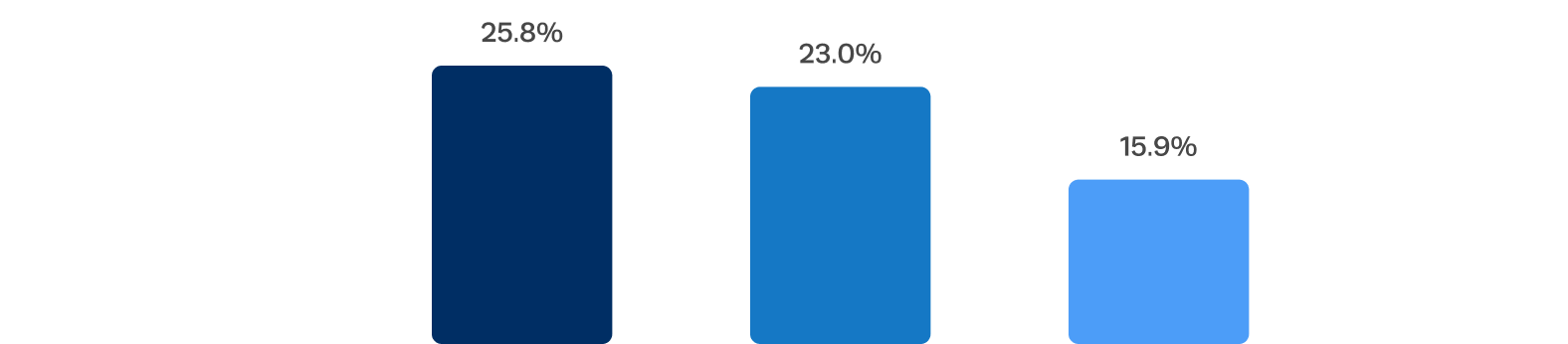

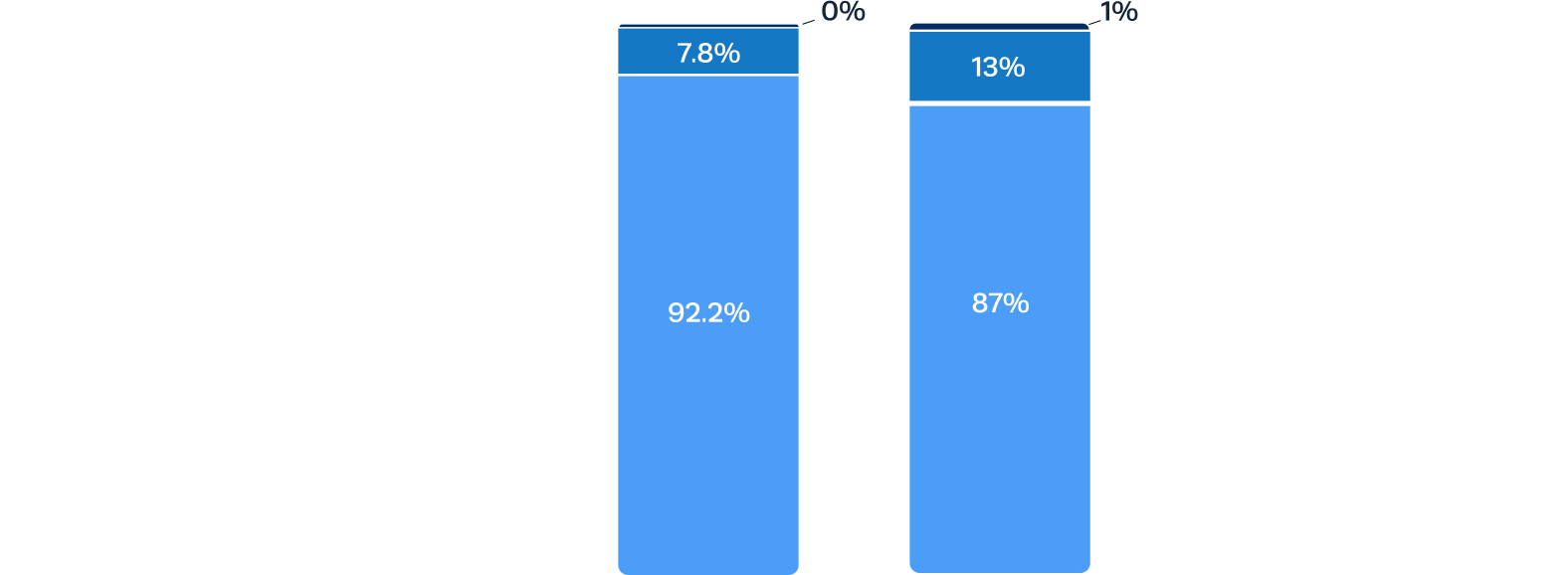
Sustained low rates of paravalvular regurgitation at 2 years
Residual aortic regurgitation is a common complication of off-label use of TAVR devices to treat AR.3 After Trilogy TAVR, significant aortic regurgitation was eliminated in all patients.
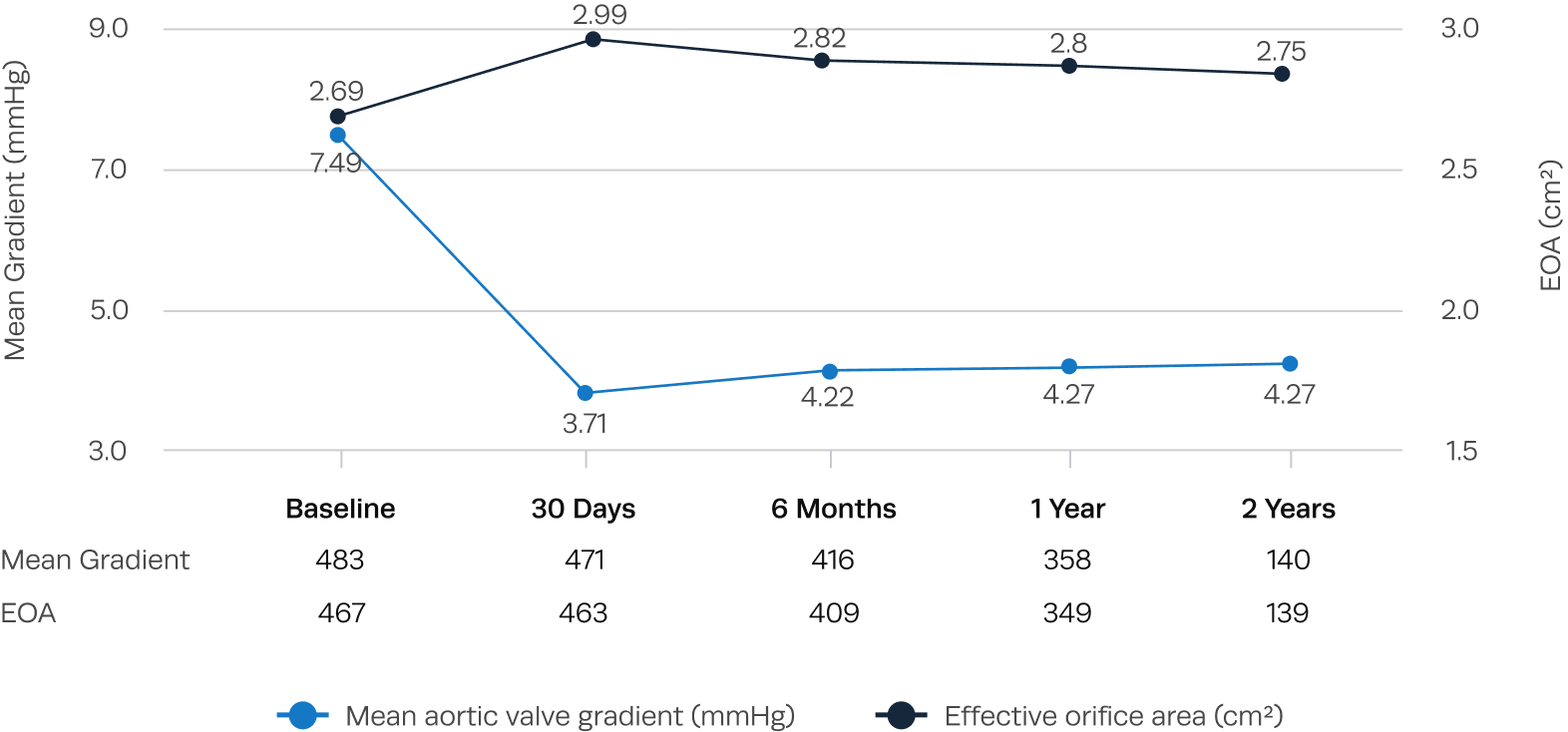
Excellent hemodynamic valve performance
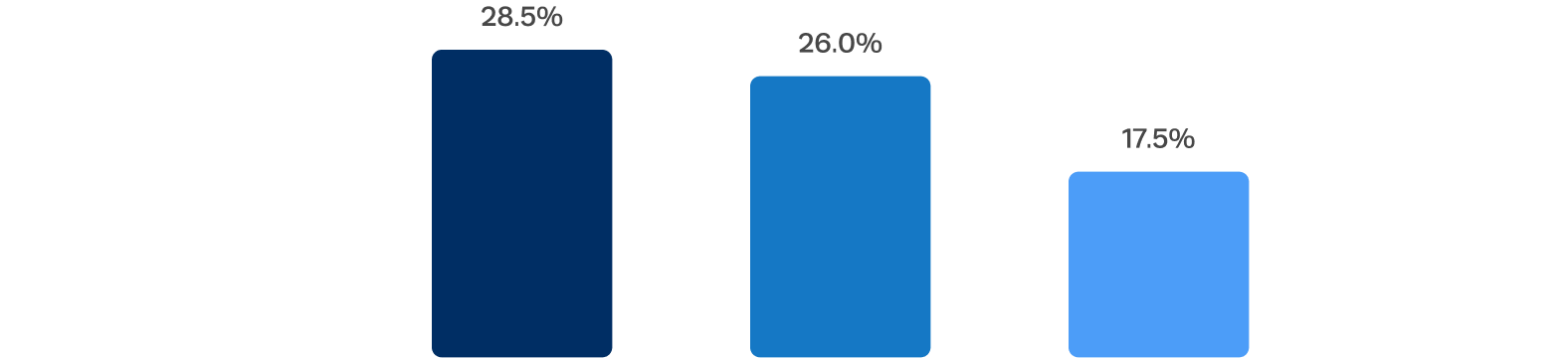
Low single-digit transvalvular gradients and large EOAs observed after Trilogy TAVR.
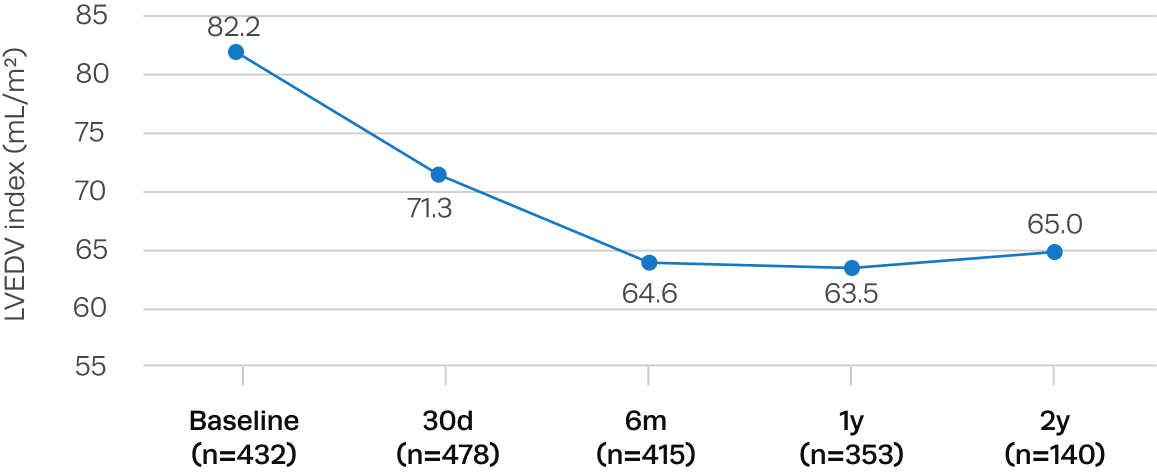
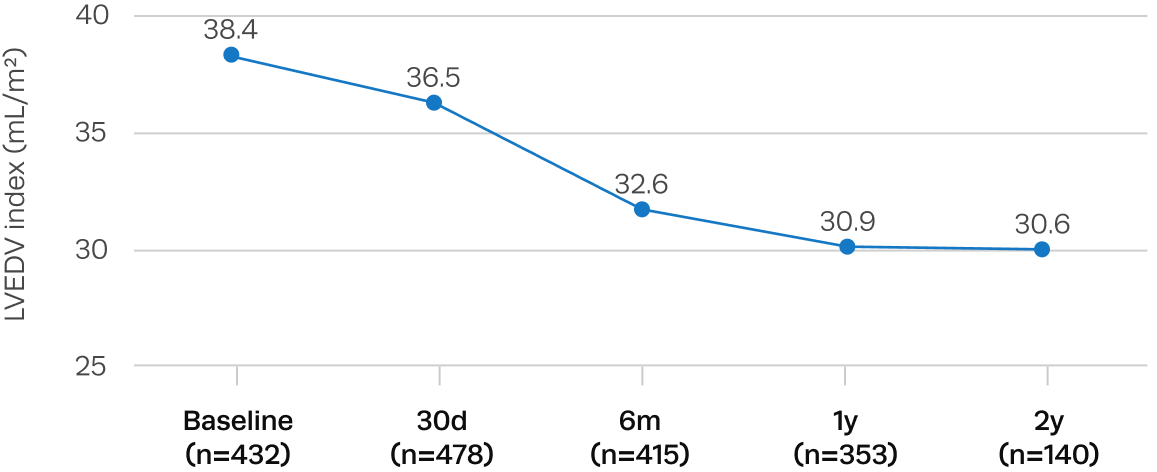
Significant left ventricular remodeling and reduction in LV mass after Trilogy TAVR
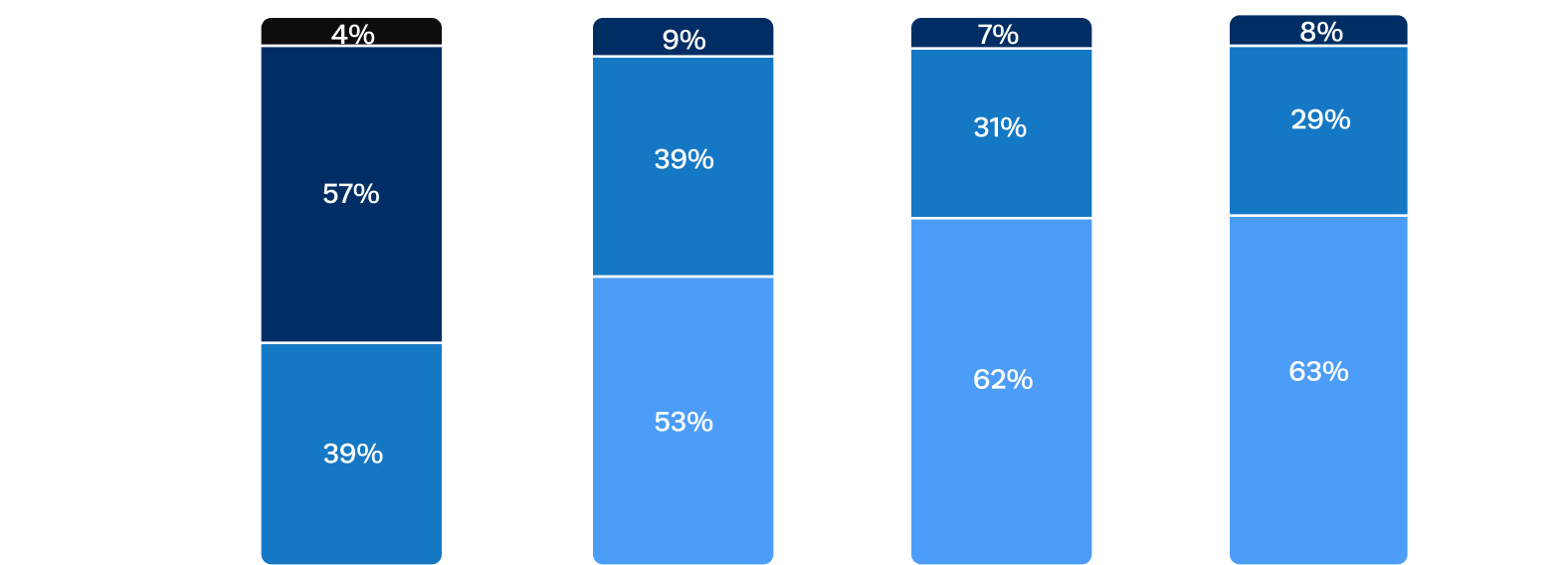
Sustained improvements in quality of life after Trilogy TAVR
At 1 year, patients experienced improvements in functional status with 92% in NYHA I/II, compared to 68% of patients in NYHA III/IV at baseline.
Watch the full late-breaking
presentation from TCT
Please complete the contact form to download the ALIGN-AR Study presentation slides
1. Vahl TP, et al. Lancet. 2024;403-1451-1459.
2. Poletti E, et al. JACC Cardiovasc Interv. 2024;17:1597-1606.
3. Haddad A et al., Clinical Cardiology 2019;42:159-166.
*at the end of the procedure
†Composite of 30 day all-cause mortality, all stroke, life-threatening/major bleeding, major vascular complications, AKI ≥ 2 or dialysis, valve intervention, new permanent pacemaker, ≥ moderate PVR
※Patients with pre-existing pacemakers were excluded from the denominator, n=150.
§Patients with pre-existing pacemakers were excluded from the denominator, n=589.
¶Some patients did not undergo 30-day echocardiographic interpretation by the core laboratory, n=569.
‡All-cause mortality




