The only
TAVR system
FDA-approved
for aortic
regurgitation.
INTRODUCING
Trilogy
Transcatheter
Heart Valve
The valve that
makes TAVR for
AR possible
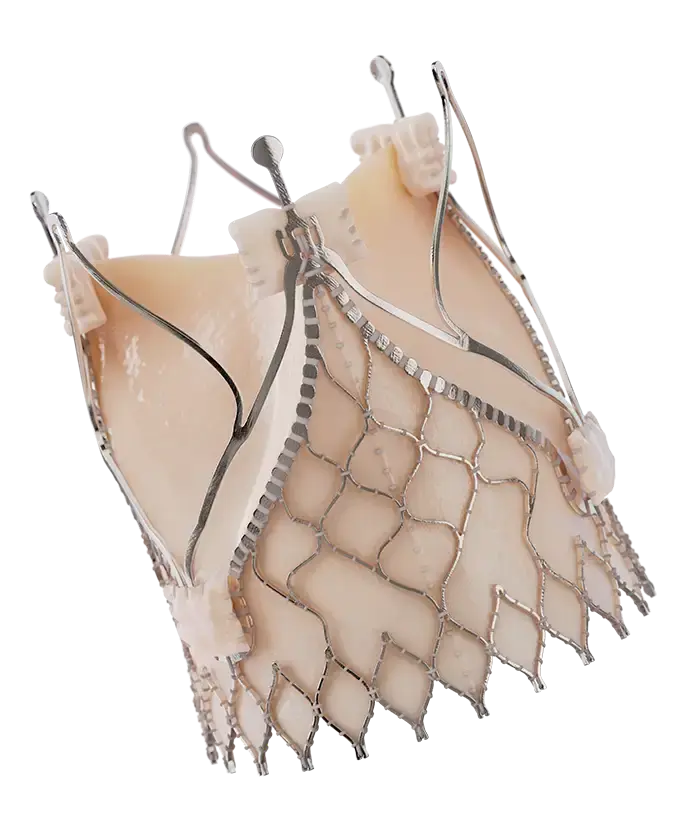


The Trilogy Valve with locator technology
You can finally give high-risk patients with severe, symptomatic aortic regurgitation (AR) an FDA-approved transcatheter aortic valve replacement (TAVR) treatment option. Trilogy’s locator technology ensures commissural alignment and allows the valve to attach to the native leaflets for secure anchoring, even in the absence of calcium.


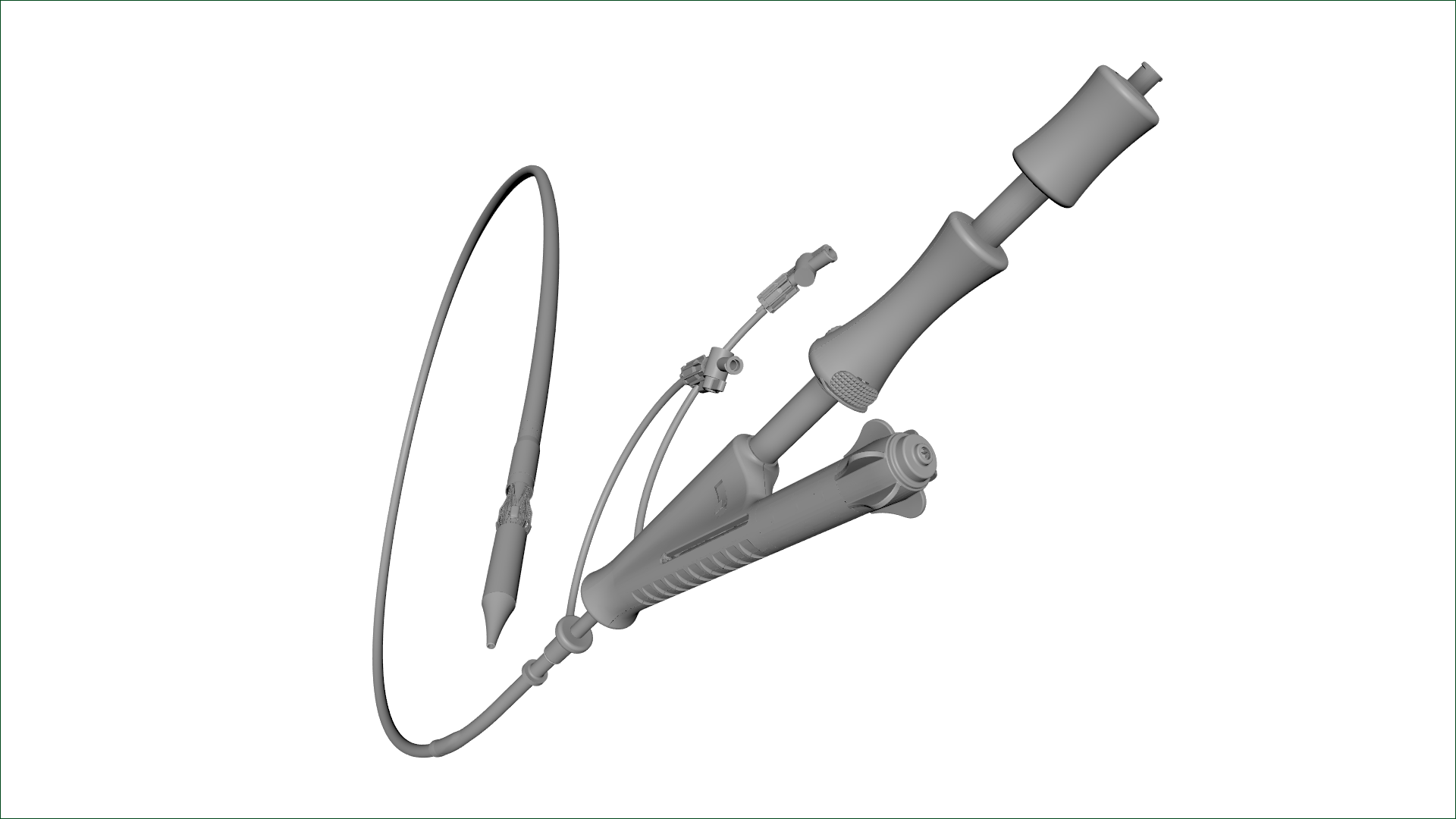
The Trilogy Delivery System
The delivery system has been engineered specifically for precise and reliable single-action valve deployment within the patient anatomy. It utilizes a catheter deflection mechanism to center the valve above the annulus and integrated rotation and axial positioning for simple and straightforward commissural alignment.


The Trilogy Introducer Sheath
Unlike other sheaths that only reach the abdominal aorta, the 85 cm pre-shaped, hydrophilically-coated Trilogy sheath extends to the sinotubular junction. This ensures the Trilogy Valve and your patient are protected all the way until release.

TAVR Procedure
A transcatheter solution for aortic regurgitation has been impossible—until now.
Compared to 10% with off-label TAVR devices3,*
Compared to 10% with off-label TAVR devices3,*
Explore Clinical Evidence From the ALIGN-AR Study
See how the Trilogy® Heart Valve System performed in the ALIGN-AR study—the first and only FDA-approved TAVR for AR.


Download device brochure
See how the Trilogy System is designed to treat severe AR patients
Download Device Brochure

1. Makkar RR, et al. The Lancet. 2025;406(10521):2757–2771.
2. Vahl TP, et al. Lancet. 2024;403-1451-1459.
3. Poletti E, et al. JACC Cardiovasc Interv. 2024:17:1597-1606.
*at the end of the procedure
Important Safety Information for the Trilogy Transcatheter Heart Valve System
Indications for use: The Trilogy® Transcatheter Heart Valve System is indicated for the treatment of symptomatic, severe native tricuspid aortic valve regurgitation (not due to acute endocarditis, rheumatic heart disease, or acute aortic dissection) in patients who are judged by a Heart Team, including a cardiac surgeon, to be at high or greater risk for surgical aortic valve replacement (i.e., predicted risk of surgical mortality ≥ 8% at 30 days, based on the Society of Thoracic Surgeons (STS) risk score and other clinical co-morbidities unmeasured by the STS risk calculator).
Contraindications: The Trilogy Transcatheter Heart Valve System is contraindicated in patients who cannot tolerate an anticoagulation/antiplatelet regimen, have known hypersensitivity to nitinol alloy (nickel and titanium) or contrast agents that cannot be managed with premedication, or who have active bacterial endocarditis or other active infection.
Warnings General: The Trilogy® THV is only to be used with the Trilogy® IS, Trilogy® DC and Trilogy® LT. Use of other devices may damage the THV and/or result in patient injury. This procedure should only be performed by physicians experienced in transcatheter aortic valve replacement (TAVR) procedures and trained in the use of the Trilogy® Transcatheter Heart Valve System, and where emergency cardiac surgery (surgical aortic valve replacement) may be promptly performed. The procedure must be conducted under fluoroscopic guidance. Some fluoroscopically guided procedures are associated with a risk of radiation injury to the skin. These injuries may be painful, disfiguring, and long-lasting. Ensure the guidewire is in proper location in the ventricle throughout the procedure to mitigate risk of ventricular wall perforation. Improper management of the guidewire, allowing it to move forward into the left ventricle, may lead to ventricular injury requiring intervention. Systemic anticoagulation with heparin should be administered and adjusted as needed throughout the procedure per hospital and physician preference. Activated clotting time (ACT) > 250 seconds is recommended to be maintained prior to Introducer Sheath insertion through completion of the procedure. Failure to maintain proper ACT levels may lead to patient harm or death. If heparin is contraindicated, an alternative anticoagulant should be considered and administered as per institutional policy. The THV must be sized appropriately to fit the patient’s anatomy. Proper device sizing is the responsibility of the treating physician. Refer to the THV sizing chart in the instructions for use. Failure to implant a device within the sizing matrix could lead to adverse effects such as those listed under the potential adverse events section. DO NOT bend or kink the Introducer Sheath. Doing so may damage the Delivery Catheter and the THV during or after the loading process. DO NOT mix the sizes of the THV, Delivery Catheter, or Loading Tools. Use of mixed size configurations of THV, Delivery Catheter, and Loading Tools may result in implantation difficulties such as premature/failure to release the THV from the Delivery Catheter, THV damage, or patient injury requiring intervention. DO NOT use the Trilogy® THV System components if: the Use-By date has elapsed; the packaging or any of its components are not sterile or have been unintentionally opened before use; or the product has been dropped, damaged, or mishandled. DO NOT use the Trilogy® IS with any pre-implant balloon valvuloplasty catheter. Trilogy THV: DO NOT use the THV if: the serial number tag is missing or does not match the container label; the container or seal is damaged, cracked, or leaking; the temperature indicator is out of range; or the THV is not fully covered in solution after opening the package. Incorrect sizing of the THV with the patient’s native aortic valve may lead to aortic leak, migration, annular injury, embolization, and/or unsuccessful implant. Accelerated deterioration of the THV may occur in patients with an altered calcium metabolism.
Precautions General: DO NOT expose the THV to solutions other than the storage, sterile rinse, and sterile chilling solutions. DO NOT add any other substance or drug to the THV storage, sterile rinse, or sterile chilling solutions. Always keep the THV tissue moist with rinsing or immersion. The safety and effectiveness of the THV has not been evaluated in the pediatric and pregnant/nursing patient population. The Trilogy® THV System has not been evaluated for use in patients with severe renal insufficiency (GFR < 30 mL/min) or renal disease requiring renal replacement therapy (e.g., dialysis). The Trilogy® THV System has not been evaluated for use in patients with percutaneous Ventricular Assist Devices (pVAD) or Left Ventricular Assist Devices (LVAD). Long-term clinical durability has not been established for the Trilogy transcatheter heart valve. Engineering testing has demonstrated a valve durability equivalent to 3.5 years in the laboratory, which did not meet the 5 years recommended by the relevant international standard. Regular check-ups with your doctor are very important. The safety and effectiveness of the THV has not been evaluated in patient populations presenting with the following: blood dyscrasias as defined: leukopenia (WBC < 3000/mm³), thrombocytopenia (platelets < 90,000/µl) or anemia (Men: Hgb < 8.1 g/dl; Women: Hgb < 7.4 g/dl); congenital/functional unicuspid, bicuspid or quadricuspid native aortic valve morphology; previous prosthetic aortic valve (bioprosthesis or mechanical) implant; echocardiographic evidence of current left heart thrombus; hypertrophic cardiomyopathy with or without obstruction; severe pulmonary hypertension (systolic PA pressure > 80 mmHg) or severe RV dysfunction as assessed clinically and by echo; very severely reduced left ventricular ejection fraction (LVEF < 25%); significant disease of ascending aorta, including ascending aortic aneurysm (defined as maximal luminal diameter of 50 mm or greater) or significant protruding or ulcerated atheroma; aortic annular diameter of less than 21 mm or more than 28.6 mm (assessed by multidetector computed tomography (MDCT) measurement); or access vessel characteristics that would preclude safe placement of the JenaValve 20Fr introducer sheath, such as severe obstructive calcification, severe tortuosity, or vessel diameter <7mm.
Prior to Use: Removal of the Delivery Catheter and Introducer Sheath from the respective packaging should be performed carefully to prevent damage or kinking. The Trilogy® THV System is designed for single patient use only. Do not reuse, reprocess or resterilize any component of this product. Reusing, reprocessing or resterilizing this product may compromise its initial integrity which could result in patient injury, illness, or death. The THV and the glutaraldehyde storage solutions are STERILE. The exterior of the THV container is NONSTERILE and must NOT be placed in the sterile field. Exposure to glutaraldehyde may cause irritation of the skin, eyes, nose, and throat. Avoid prolonged or repeated exposure to the vapors and use adequate ventilation. If skin contact occurs, immediately flush the affected area with water. In the event of eye contact, flush with water and seek immediate medical attention.
During Use: Adequate rinsing of the THV with sterile cold saline prior to use as described in this manual is mandatory before implantation. No other solutions, drugs, or chemicals (including antibiotics) should be added to the storage or rinsing solutions, as irreparable damage to the THV Leaflets, which may not be apparent under visual inspection, may occur. The use of this device requires administration of intravascular contrast. Patients with pre-existing renal insufficiency may have an increased risk of renal failure post-operatively (e.g., patients with Stage 4 or 5 chronic kidney disease). Care should be taken to limit the amount of contrast media used during the procedure. Introducer Sheath and Delivery Catheter advancement should be performed under fluoroscopic guidance. Do not use excessive force to advance or withdraw the Introducer Sheath or Delivery Catheter when resistance is encountered. Vessel or device damage may occur. Care should be taken in areas of stenosis, intravascular thrombosis, or in calcified and/or tortuous vessels. Pre-dilatation of the native aortic annulus using balloon aortic valvuloplasty (BAV) is performed at physician discretion. Ensure guidewire access and positioning is maintained at all times. Do not remove the guidewire while the Delivery Catheter and Introducer Sheath are inserted in the patient. Inaccurate placement, inadequate Locator to Leaflet fixation and/or incomplete sealing of the THV within the annulus may result in increased risk of aortic leakage, migration, or inadvertent coronary occlusion. Incorrect deployment or migration of the THV may require intervention. Once deployment has occurred, repositioning of the THV (e.g., using a snare) is not recommended. Repositioning of a deployed THV may cause patient injury and/or require emergent surgery. DO NOT attempt to retrieve or recapture the prosthetic aortic valve if the sealing ring has been deployed. If the sealing ring has been deployed, the THV must be released from the catheter before the catheter can be withdrawn with the Introducer Sheath. Take care during manipulation of catheters, wires, and sheaths within the aorta. Excessive manipulation may dislodge fragments of atheroma or calcification which can cause embolization. Clinical long-term durability for the Trilogy® THV has not been established. Regular medical follow-up is advised to evaluate THV performance as per standard of care for similar bio-prosthetic valves. Post-procedure, administer appropriate antibiotic prophylaxis as needed for patients at risk of prosthetic valve infection or endocarditis. Post-procedure, administer anticoagulation and/or antiplatelet therapy per physician judgment.
Potential Adverse Events
Below is a list of the potential adverse effects (e.g., complications) associated with the use of the device. Death; allergic reaction to anesthesia, contrast media, antithrombotic therapy, device materials; anemia; angina; aortic root distortion; atelectasis; arrhythmia; arteriovenous (AV) fistula; blood loss requiring transfusion; cardiovascular or vascular injury, such as perforation or damage (dissection) of vessels, myocardium or valvular structures that may require intervention; cardiac arrest; cardiac failure; cardiogenic shock; chest pain/discomfort; conduction system injury; coronary flow obstruction/transvalvular flow disturbance; deep vein thrombosis; device acute migration or malposition; device dysfunction (regurgitation and/or stenosis); device embolization; device thrombosis; dislodgement of previously implanted devices (i.e., pacing lead); dyspnea; electrolyte imbalance; embolic event: air, calcific material, thrombus, device fragments; endocarditis; exercise intolerance or weakness; fever; hematoma or ecchymosis; hemolysis/hemolytic anemia; hypertension or hypotension; infection including incisional site infection, septicemia and endocarditis; inflammation; mechanical failure of delivery system, and/or accessories; myocardial infarction; pain; pericardial effusion/cardiac tamponade; pleural effusion; pneumothorax; pulmonary edema; radiation injury; renal insufficiency or renal failure; reoperation; respiratory insufficiency or respiratory failure; stroke/transient ischemic attack; syncope; systemic or peripheral ischemia; systemic or peripheral nerve injury.
CAUTION: Federal (USA) law restricts these devices to sale by or on the order of a physician. Implantation of the transcatheter heart valve should be performed only by physicians who have received JenaValve Technology Inc. training. Indications, contraindications, warnings, and instructions for use can be found in the product labeling supplied with each device. For more information please refer to https://eifu.jenavalve.com



 Scroll to explore
Scroll to explore 

 Menu
Menu  Close
Close